 ISBN 978-0-08-037941-8.Trending Questions What is neodymium's state of matter at room temperature? When atoms are drawn together by attractive forces their potential energy is what? How many grams of CH3OH must be added to water to prepare 150 mL of a solution that is 1. Cesium ion (1+) Formula: Cs + Molecular weight: 132.9049033 IUPAC Standard InChI: InChI1S/Cs/q+1 Copy. Like sodium oxide, calcium nitride absorbs hydrogen above 350 ☌:Ĭa 3N 2 + 2 H 2 → 2 CaNH + CaH 2 General references 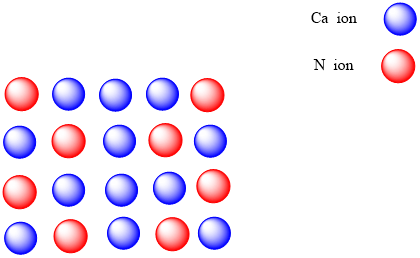
It reacts with water or even the moisture in air to give ammonia and calcium hydroxide: Ca 3N 2 + 6 H 2O → 3 Ca(OH) 2 + 2 NH 3 It can be produced by direct reaction of the elements: 3 Ca + N 2 → Ca 3N 2 textbook question: Write formulas for the Ionic compounds formed by the following ions. Synthesis and reactions Ĭalcium nitride is formed along with the oxide, CaO, when calcium burns in air. In this structure, Ca 2+ occupies tetrahedral sites, and the nitride centres occupy two different types of octahedral sites. Α-Calcium nitride adopts an anti- bixbyite structure, similar to Mn 2O 3, except that the positions of the ions are reversed: calcium (Ca 2+) take the oxide (O 2−) positions and nitride ions (N 3−) the manganese (Mn 3+). What is in cesium sulfate Caesium sulfate or cesium sulfate is the inorganic compound and salt with the formula Cs2SO4. Is cesium nitrate an acid or base cesium nitrite is the salt of a weak acid which is a generic term for hundreds of salts of this type. Strontium nitride, Sr 3 N 2, is produced by burning strontium metal in air (resulting in a mixture with strontium oxide) or in nitrogen. Simple Structure Advanced History Comment on this record 3D Caesium nitrate Molecular Formula CsNO Average mass 194.910 Da Monoisotopic mass 194. It exists in various forms ( isomorphs), α-calcium nitride being more commonly encountered. CsNO3 Caesium nitrate/Formula What is CsNO2 Caesium nitrite CsNO2 PubChem.  
If it is ionic, write the symbols for the ions involved: (a) NF 3. It exists in various forms (isomorphs), but -calcium nitride is more commonly. For each of the following compounds, state whether it is ionic or covalent. Calcium nitride is an inorganic compound with the chemical formula Ca3N2. Calcium nitride is the inorganic compound with the chemical formula Ca 3 N 2. Using the periodic table, predict whether the following chlorides are ionic or covalent: SiCl 4, PCl 3, CaCl 2, CsCl, CuCl 2, and CrCl 3. Ionic Formula Name of Ionic Compound Al 2 O 3 Aluminum Oxide Ba 3 N 2 barium nitride 3 Cs 2 O Cesium Oxide CaO calcium oxide Li 2 O lithium oxide Rb 3 N. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
